Arcturus Therapeutics & Duke-NUS Received Approval to Proceed with Phase 1/2 Clinical Trial for COVID-19 Vaccine Candidate, LUNAR-COV19
Human dosing of LUNAR-COV19 expected soon
Differentiated STARR™ mRNA vaccine expected to produce humoral and cellular immunity at very low doses
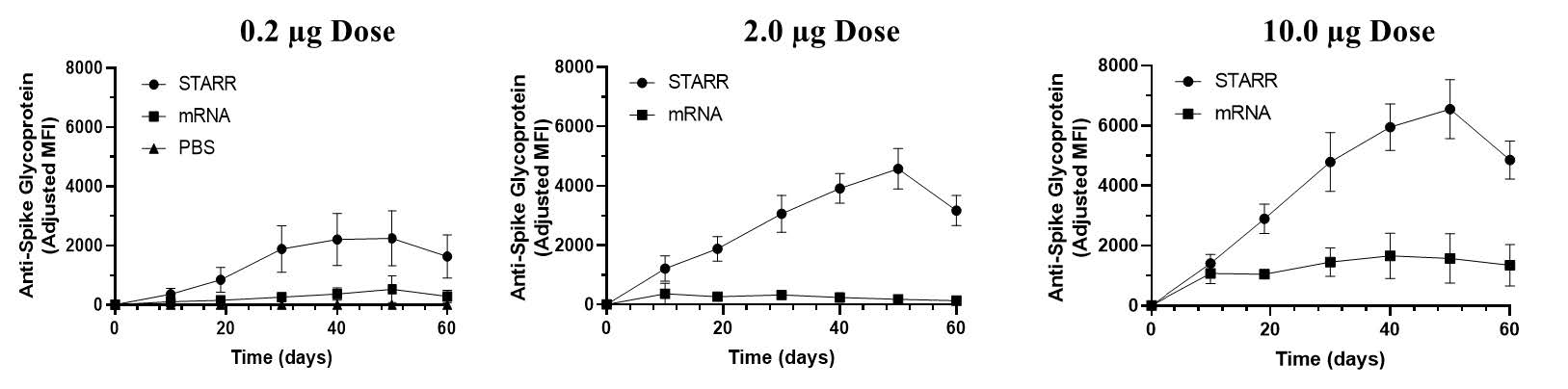
New preclinical data demonstrates neutralizing antibody titers continue to increase for 50 days after a single administration
Arcturus & Duke-NUS will initiate human dosing of LUNAR-COV19 as soon as possible. The healthy volunteer study will evaluate several dose levels of LUNAR-COV19 in up to 108 adults, including older adults. Follow-up will be conducted to evaluate safety, tolerability and the extent and duration of the humoral and cellular immune response.
“The approval of the Clinical Trial Application for LUNAR-COV19 is a critical milestone for Arcturus. We are excited to advance this promising vaccine candidate into clinical trials. Based on our preclinical data, we believe that our self-replicating mRNA-based approach may produce high rates of seroconversion and robust T-cell induction with a potential single administration, at very low doses. The LUNAR-COV19 profile is meaningfully differentiated and may facilitate the mass vaccine campaigns necessary to target hundreds of millions of individuals globally,” said
Professor
“There is a tremendous global imperative to develop effective preventive measures for COVID-19 infections. We are heartened by the rapid and promising progress in our vaccine collaboration with Arcturus as we move forward into clinical trials,” said Professor
The STARR™ Technology platform employed in LUNAR-COV19 combines self-replicating mRNA with LUNAR®, a proprietary nanoparticle delivery system optimized for mRNA molecules. The efficiency and self-replicating nature of the approach were designed to enable very low doses, and a potential single vaccine administration. Prior animal data has demonstrated robust humoral and cellular immunity elicited at doses as low as 0.2 µg of LUNAR-COV19. Additionally, Arcturus demonstrated 100% seroconversion for anti-SARS-CoV-2 neutralizing antibodies with a very low single dose (2.0 µg).
New preclinical data demonstrate that neutralizing antibody levels in response to a single administration of LUNAR-COV19 (0.2, 2.0, 10.0 µg) continue to increase over 50 days. The increasing antibody levels are attributed to the self-replicating mRNA of LUNAR-COV19. These results were obtained using a Luminex bead assay. A 1/2000 serum dilution was assayed for neutralizing IgG antibodies in the mouse serum every 10 days for 60 days post vaccination.
|
||||
About Arcturus Therapeutics
Founded in 2013 and based in
About Duke-NUS Medical School
Duke-NUS is Singapore’s flagship graduate entry medical school, established in 2005 with a strategic, government-led partnership between two world-class institutions: Duke University School of Medicine and the National University of Singapore (NUS). Through an innovative curriculum, students at Duke-NUS are nurtured to become multi-faceted ‘Clinicians Plus’ poised to steer the healthcare and biomedical ecosystem in Singapore and beyond. A leader in ground-breaking research and translational innovation, Duke-NUS has gained international renown through its five signature research programmes and eight centres. The enduring impact of its discoveries is amplified by its successful Academic Medicine partnership with Singapore Health Services (SingHealth), Singapore’s largest healthcare group. This strategic alliance has spawned 15 Academic Clinical Programmes, which harness multi-disciplinary research and education to transform medicine and improve lives. For more information, please visit www.duke-nus.edu.sg
Forward Looking Statements
This press release contains forward-looking statements that involve substantial risks and uncertainties for purposes of the safe harbor provided by the Private Securities Litigation Reform Act of 1995. Any statements, other than statements of historical fact included in this press release, including those regarding the Company’s expected performance, the Company’s development of any specific novel mRNA therapeutics, the Company’s efforts to develop a vaccine against COVID-19, and therapeutic potential thereof, based on the Company’s mRNA therapeutics, the forecasted safety, efficacy, characteristics or reliability of a vaccine against COVID-19, were one to be successfully developed based on the Company’s mRNA therapeutics, the dosing level and frequency of a vaccine against COVID-19 were one to be successfully developed based on the Company’s mRNA therapeutics and the impact of general business and economic conditions are forward-looking statements. Arcturus may not actually achieve the plans, carry out the intentions or meet the expectations or projections disclosed in any forward-looking statements such as the foregoing and you should not place undue reliance on such forward-looking statements. Such statements are based on management’s current expectations and involve risks and uncertainties, including those discussed under the heading "Risk Factors" in Arcturus’ Annual Report on Form 10-K for the fiscal year ended
Contact
(858) 900-2682
IR@ArcturusRx.com
Kendall Investor Relations
(617) 914-0008
ctanzi@kendallir.com
(+65) 6516-1138
lekshmy_sreekumar@duke-nus.edu.sg
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/fb3fb0a3-1978-4788-8811-57c446b49588
Source: Arcturus Therapeutics Holdings Inc.
Source: Duke-NUS Medical School