Arcturus Reports Additional Supportive Preclinical Data for its COVID-19 Vaccine Candidate (LUNAR-COV19)
New data provides evidence of adaptive cellular (CD8+ cells) and balanced (Th1/Th2) immune responses
STARR™ mRNA elicits anti-spike protein antibodies (IgG), higher than conventional mRNA at all doses
These scientific data supporting robust immunogenicity of LUNAR-COV19 were measured by investigators at the
“These extended preclinical studies, conducted in partnership with Arcturus, establish a comprehensive and highly compelling package of data to support clinical trials,” said Professor
Pad Chivukula, Ph.D., Chief Scientific Officer of
An additional study evaluating cell-mediated immunogenicity has been recently completed at Professor Ooi Eng Eong’s lab at Duke-NUS. The results showed a dose dependent CD8+ T-cell response, with a clear response observed at all doses, as well as a balanced Th1/Th2 CD4+ T-cell response (intracellular cytokine (IFN-γ/IL-4) staining). The percent of CD8+ T-cells increased from the 4% baseline to 8% with increasing doses of STARR™ mRNA. The Th1/Th2 ratio for T-helper cells (CD4+) shows a strong TH1 response which does not change with increasing dose, indicating that the immune response remains balanced across all dose levels.
STARR™ mRNA (LUNAR-COV19) Induces Adaptive Cellular Immunity
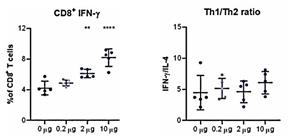
| Single Dose (µg) |
LUNAR-COV19 (STARR™ mRNA) After 7 Days | |
| % CD8+ T-Cells | Th1/Th2 | |
| 0 | 4.0 | 4.6 |
| 0.2 | 4.5 | 5.3 |
| 2 | 6.0 | 5.0 |
| 10 | 8.0 | 6.0 |
A single administration of LUNAR-COV19 STARR™ mRNA induced a higher anti-spike protein IgG response than conventional mRNA at equivalent doses, and particularly at lower doses. The IgG response also continued to increase at a much greater rate over the 30-day post vaccination period than conventional mRNA.
Immunogenicity1 (Anti-spike protein IgG) – STARR™ mRNA vs. Conventional
| Single Dose (µg) |
LUNAR® Delivery | |||||
| STARR™ mRNA (MFI)2 | Conventional mRNA (MFI)2 | |||||
| Day 10 | Day 19 | Day 30 | Day 10 | Day 19 | Day 30 | |
| 0.2 | 361 | 850 | 1887 | 110 | 154 | 260 |
| 2 | 1217 | 1885 | 3062 | 367 | 267 | 327 |
| 10 | 1413 | 2895 | 4789 | 1077 | 1053 | 1456 |
1Immunogenicity is defined as the induction of multiple antibodies (IgG) that bind to the spike protein following intramuscular administration.
2 Mean Fluorescence Intensity (MFI); Serum diluted 1 to 2000.
A vaccine approach that elicits a broad and balanced immune response by activating both humoral and cellular immunity has the potential to provide more effective protection. An important advantage of our self-replicating mRNA vaccine is the potential to activate both of these important components of adaptive immunity and this preclinical LUNAR-COV19 data confirms a robust and balanced immune response.
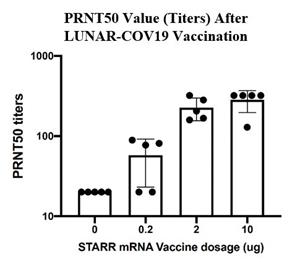
Seroconversion was determined using a quantitative plaque reduction neutralization assay (using SARS-CoV-2 Singapore Clinical Isolate), which measured neutralizing antibody titers are detected in serum at day 30. Rodents were immunized with a single intramuscular dose (0.2, 2, and 10 µg) of LUNAR-COV19 vaccine.
At day 30 post vaccination, 80% of mice vaccinated with 0.2 µg LUNAR-COV19 elicited antibodies that neutralized 50% of SARS-CoV-2 at titers 20 and above. The geometric mean titer of the 4 animals with titers >20 is 57.72 (SD = 2.032). At the same time point, 100% animals vaccinated with 2 µg of LUNAR-COV19 developed antibody titers >20, with geometric mean titer of 217.9 (SD = 1.365). The 10 µg dose of LUNAR-COV19 vaccine produced titers of 320 or greater in 80% of animals, which was the upper limit of dilution of this test. The concentration of serum to reduce the number of plaques by 50% compared to the serum free virus gives the measure of how much antibody is present or how effective it is. This measurement is denoted as the PRNT50 value. The plaque reduction neutralization test is used to quantify the titer of neutralizing antibody for a virus.
About
Founded in 2013 and based in
About STARR™ Technology
The STARR™ Technology platform combines self-replicating mRNA with LUNAR®, a leading nanoparticle delivery system, into a single solution to produce proteins inside the human body. The versatility of the STARR™ Technology affords its ability upon delivery into the cell to generate a protective immune response or drive therapeutic protein expression to potentially prevent against or treat a variety of diseases. The self-replicating RNA-based therapeutic vaccine triggers rapid and prolonged antigen expression within host cells resulting in protective immunity against infectious pathogens. This combination of the LUNAR® and STARR™ technology is expected to provide lower dose requirements due to superior immune response, sustained protein expression compared to non-self-replicating mRNA-based vaccines and potentially enable us to produce vaccines more quickly and simply.
Forward Looking Statements
This press release contains forward-looking statements that involve substantial risks and uncertainties for purposes of the safe harbor provided by the Private Securities Litigation Reform Act of 1995. Any statements, other than statements of historical fact included in this press release, including those regarding the Company’s expected performance, the Company’s development of any specific novel mRNA therapeutics, the Company’s efforts to develop a vaccine against COVID-19 based on the Company’s mRNA therapeutics, the forecasted safety, efficacy or reliability of a vaccine against COVID-19, were one to be successfully developed based on the Company’s mRNA therapeutics, the timing and availability of a vaccine against COVID-19 were one to be successfully developed based on the Company’s mRNA therapeutics, the potential initiation of human trials of a vaccine against COVID-19 based on the Company’s mRNA therapeutics, the timing of initiation of human trials of a vaccine against COVID-19 based on the Company’s mRNA therapeutics, the potential market impact of a vaccine against COVID-19 based on the Company’s mRNA therapeutics and the impact of general business and economic conditions are forward-looking statements. Arcturus may not actually achieve the plans, carry out the intentions or meet the expectations or projections disclosed in any forward-looking statements such as the foregoing and you should not place undue reliance on such forward-looking statements. Such statements are based on management’s current expectations and involve risks and uncertainties, including those discussed under the heading "Risk Factors" in Arcturus’ Annual Report on Form 10-K for the fiscal year ended
Contact
(858) 900-2682
IR@ArcturusRx.com
Kendall Investor Relations
(617) 914-0008
ctanzi@kendallir.com
Photos accompanying this announcement are available at
https://www.globenewswire.com/NewsRoom/AttachmentNg/06a2e8fe-5e76-47e0-897e-7303baeacf62
https://www.globenewswire.com/NewsRoom/AttachmentNg/3879704c-a063-4570-aa96-2e44a1341a6d
Source: Arcturus Therapeutics Holdings Inc.
